|
There was no family history of similar eye disturbance. She denied using illicit drugs, herbal, or other medications, including ones over the counter. Her medications at presentation were duloxetine HCl 60 mg daily, trazodone HCl 100 mg at night, gabapentin 900 mg three times a day, and clonazepam 0.5 mg twice a day as needed. Her past medical history was important for obesity, tobacco use, fibromyalgia, and depression. She denied any recent head trauma, fever, chills, or other complaints. 
Severe episodes were sometimes associated with transient right eye mydriasis ( Figure 1), significant lower extremity weakness and at times confusion that all resolved with the resolution of her migraine episode. This is an abstract that was submitted for the 2017 ARVO Annual Meeting, held in Baltimore, MD, May 7-11, 2017.A 30-year-old lady with a history of migraine headache presented with a one-year history of chronic intermittent, bilateral, throbbing, and at times very severe, headache that often lasted for days and was associated with blurry vision, nausea, vomiting, photophobia, and phonophobia. If confirmed by prospective clinical trials, these findings would offer novel treatments for myopia progression. Any cycloplegic effect, while not detected in this patient, needs to be further investigated in the young myopic population. That the pupil remained reactive to changes in ambient light, rather than fixed and dilated as with standard atropine eye drop dosing, we believe contributes to the comfort of such treatment. Dosing remained low enough to continuously prevent symptoms of blur and light sensitivity. the control eye at every measurement over 60 days. 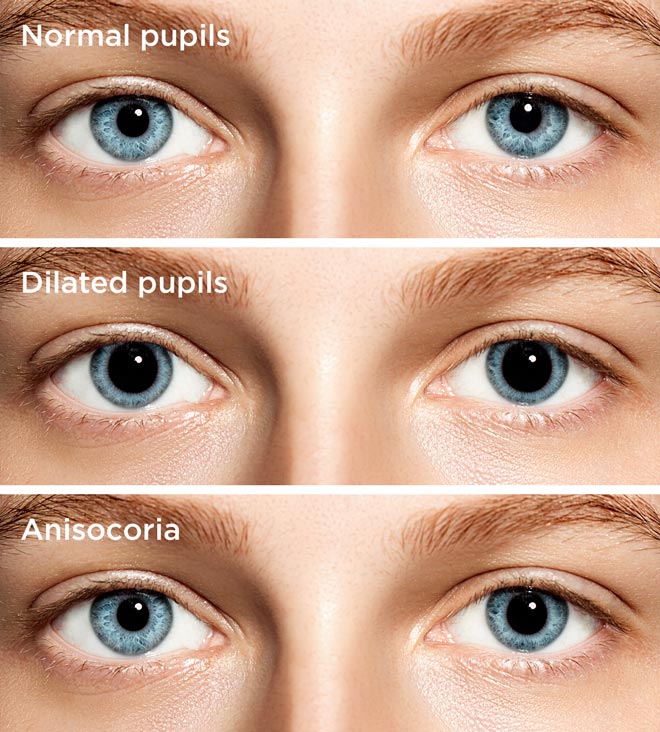
The study established intraocular continuous very low dose atropine delivery, as demonstrated by an increased pupil size vs. After moderate blur and mild glare the first evening of treatment only, there were no visual symptoms of light sensitivity or blur throughout the study. The 0.05% dose did not fix and dilate the pupil at any time during treatment. Lighting conditions were varied to confirm the treated pupil’s constriction and dilation function.įor the entire time that the device was worn, the treated eye had a clinically detectable larger pupil compared to the eye with no treatment for all measurements (avg 1.50 mm through 60 days, p<<.001, paired t-test). 
Pupil measurements were taken a least daily using a Jaeger Nearpoint Vision Card’s pupil gauge. The other eye functioned as the untreated control. The subject, CL, inserted one device in the left eye and wore it continuously for over 60 days. Preliminary experiments indicated trial of a hundred-fold decrease in concentration from that which caused a typical clinical drop result of a fixed dilated pupil lasting over a week. We explored whether sustained, micro quantities of atropine could be released from a topical matrix device, as evidenced by continuous but modest pupil dilation, with preserved pupil functioning and minimal light sensitivity and visual blur. The most effective concentrations cause excessive, fixed pupillary dilation, photophobia, near vision blur and possible toxic light levels to the retina, presenting obstacles to this treatment. Atropine drops have been used in the treatment of myopia progression.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
